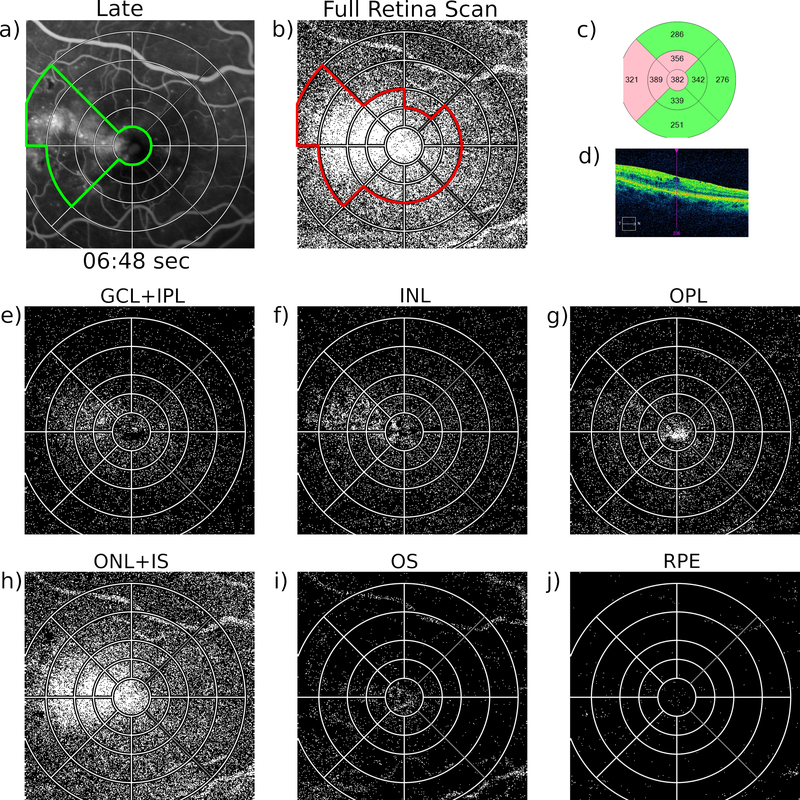
Last March 18, the European Medicines Agency (EMA) announced the selected consortium to monitor possible adverse events to COVID-19 vaccines in Europe for the next 3 years.
The consortium, led by the Netherlands, includes 13 countries and the Portuguese participation is coordinated by UFC, leaded by Prof. Francisco Batel Marques. Entitled “Monitoring the safety of covid-19 vaccines in the EU”, the study has the contribution of Portugal, through a collaboration agreement between the pharmacovigilance units of Coimbra (AIBILI-UFC and Faculty of Pharmacy of UC), Porto (Faculty of Medicine) and Lisbon (Faculty of Pharmacy).
“It is very important, from the point of view of public health, to have reliable information on the safety of vaccines”, said Prof. Francisco Batel Marques, observing also that the time of research and development was relatively short.
The project is now in preparation to start in agreement with all details approved by EMA.
More information: https://expresso.pt/coronavirus/2021-03-22-Covid-19.-Portugal-integra-projeto-europeu-para-fiscalizar-seguranca-das-vacinas-1c594763